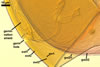
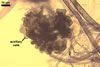
GERMINATION
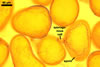
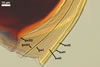
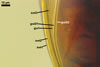
SHIELD ellipsoid to irregular; hyaline to pale yellow (1A4);
70-85 x 115-137 µm; with deep folds partitioning 5-7 lobes with smooth
margins, formed on germinal wall 2.
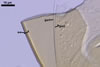
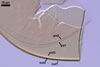
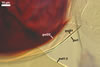
AUXILIARY
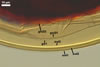
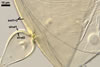
CELLS rarely single in the soil, usually in loose clusters
of 2-8; hyaline to pale yellow (2A2); pyriform to irregular; 22.5-32.5 x 37.5-47.5
µm; produced on coiled hyphae, 2.5-6.5 µm diam, concolourous with
auxiliary cells.
MYCORRHIZAE.
Associated in the field with roots of, e. g., Agrostis capilaris (Griffioen 1994), Allium porrum, Ammophila arenaria, Anthriscus silvestris, Artemisia campestris, Crataegus monogyna, Festuca polesica, F. rubra, Fragaria vesca, Glycine hispida, Juniperus communis, Medicago sativa, Nicotiana tabacum, Thuja occidentalis, Triticum aestivum and Zea mays (Blaszkowski 1993a, b, 1994a, b; Blaszkowski et al. 2002; Blaszkowski, unpubl. data; Iwaniuk and Blaszkowski 2004a, b; Tadych and Blaszkowski 2000a, b), Festuca arundinacea , F. rubra , Lotus corniculatus , and Trifolium pratense (Morton and Koske 1988), Hyacinthoides non-scripta (Merryweather and Fitter 1998), and Phaseolus vulgaris (Estrada-Torres et al. 1992).
Formed arbuscular mycorrhizae in one-species pot cultures with Festuca arundinacea, Sorghum sudanense, and Trifolium pratense as the host plants (Morton and Koske 1988).
PHYLOGENETIC POSITION.
Unknown.
DITRIBUTION. Its holotype selected from spores isolated from a pot culture with Sorghum sudanense as the host plant and established from spores originally collected under Festuca arundinacea growing in Randolph Co., near Elkins, West Virginia, U.S.A. (Morton and Koske 1988). Also found in cultivated and uncultivated soils of, e. g., Canada (Marcel et al. 1988), Mexico (Estrada-Torres et al. 1992), United Kingdom (Merryweather and Fitter 1998), the Netherlands (Griffioen 1994), and Hungary (Takacs and Bratek 2006). One of the most frequently found species of arbuscular fungi in different soils of Poland, especially in dune sites, and the most frequently occurring Scutellospora sp. in this country (Blaszkowski 1993a, b, 1994a, b; Blaszkowski et al. 2002; Blaszkowski, unpubl. data; Iwaniuk and Blaszkowski 2004a, b; Tadych and Blaszkowski 2000a, b).
NOTES. The species most similar to S. dipurpurescens is S. calospora. The two fungi are indistinguishable when observed under a dissecting microscope: their spores are identical in colour, shape, and size (Blaszkowski 1989; Franke and Morton 1994; Koske and Walker 1986; Morton 2000; Nicolson and Schenck 1979). They differ only in the structure of the germinal wall 1. In S. dipurpurescens, this wall is 1-layered, whereas the germinal wall 1 of S. calospora spores includes two layers.
REFERENCES
Blaszkowski J. 1989.
Polish Endogonaceae. I. Acaulospora bireticulata, Entrophospora
infrequens, Glomus caledonium, and Scutellispora pellucida.
Karstenia 29, 1-10.
Franke M., Morton J.
B. 1994. Ontogenetic comparisons of arbuscular mycorrhizal fungi Scutellospora
heterogama and Scutellospora pellucida: revision of taxonomic
character concepts, species descriptions, and phylogenetic hypotheses. Can.
J. Bot. 72, 122-134.
Koske R. E., Walker
C. 1986. Species of Scutellospora (Endogonaceae) with smooth-walled
spores from maritime sand dunes: two new species and a redescription of the
spores of Scutellospora pellucida and Scutellospora calospora.
Mycotaxon 27, 219-235.
Morton J. B. 2002. International
Culture Collection of Arbuscular and Vesicular-Arbuscular Mycorrhizal Fungi.
West Virginia University. http://www.invam.caf.wvu.edu/.
Nicolson T. H., Schenck
N. C. 1979. Endogonaceous mycorrhizal endophytes in Florida. Mycologia 71,
178-198.