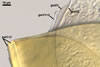
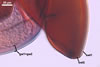
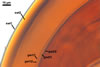
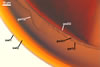
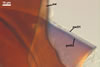
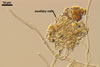
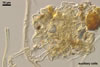
GERMINATION
SHIELD ellipsoid; hyaline to pale yellow (4A3); 65-80 x 100-120
µm; with deep folds partitioning 4-7 lobes with smooth margins; formed
on germination wall 2.
AUXILIARY
CELLS rarely single in the soil, usually in loose clusters of
2-10; hyaline to pale yellow (4A3); pyriform to irregular; 30.0-42.0 µm;
produced on coiled hyphae, 2.2-5.8 µm diam, concolorous with auxiliary
cells.
MYCORRHIZAE.
Spores of S. pellucida have been found associated
in the field with many cultivated and uncultivated plant species (Blaszkowski
1989, 1993a, b, 1994; Blaszkowski et al. 2002; Tadych and Blaszkowski 2000).
Attempts to establish one-species cultures of this fungus failed.
DISTRIBUTION.
In
Poland, S. pellucida has been widely distributed in both cultivated
and uncultivated soils (Blaszkowski 1989, 1993a, b; Iwaniuk and Blaszkowski 2004). However, this fungus has generally occurred infrequently. Most of
the S. pellucida specimens collected in Poland came from dune soils,
including those of the Hel Peninsula (54o47’N, 18o25’E-54o36’N,
18o49’E; Blaszkowski 1994), the Gdansk and Szczecin coasts (Blaszkowski
1993b), Slowinski National Park (54o45’N, 17o26’E; Tadych and
Blaszkowski 2000), and the Bledowska Desert (50o22’N, 19o34’E;
Blaszkowski et al. 2002).
Scutellospora
pellucida occurs in the whole world. This fungus has originally been
described from spores isolated from a soybean-rhizosphere soil collected in
Florida (Nicholson and Schenck 1979). Later, spores of S. pellucida have
been found, e. g., in other states of the USA (Hetrick and Bloom 1983;
Koske 1987; Miller
et al. 1985; Rose 1988), Italy (Giovannetti 1985), Israel and Turkey (Blaszkowski
et al. 2001; Blaszkowski, pers. observ.).
NOTES.
When observed under a dissecting microscope, mature spores of S. pellucida
most resemble those of S. fulgida due to their similar size and colour
(Koske and Walker 1986; Morton 1995). Older and field-collected spores of S.
pellucida are also reminiscent of light-coloured spores of S.
calospora (Koske and Walker 1986; Morton 2000; Nicolson and Gerdemann
1968; Nicolson and Schenck 1979).
These fungi differ in
the subcellular structure of spores and the biochemical properties of its
components. The subcellular structure of spores of S. pellucida and
S. calospora is identical, whereas spores of S. fulgida
lack either the third spore wall layer and the germination wall 2 of the two
former species (Morton 1995, 2000). Additionally, the laminate spore wall
layer of only S. pellucida spores stains red in Melzer’s reagent.
REFERENCES
Blaszkowski J. 1989.
Polish Endogonaceae. I. Acaulospora bireticulata, Entrophospora
infrequens, Glomus caledonium, and Scutellispora pellucida.
Karstenia 29, 1-10.
Blaszkowski J. 1993a.
Comparative studies of the occurrence of arbuscular fungi and mycorrhizae
(Glomales) in cultivated and uncultivated soils of Poland. Acta Mycol. 28,
93-140.
Blaszkowski J. 1993b.
The occurrence of arbuscular fungi and mycorrhizae (Glomales) in plant communities
of maritime dunes and shores of Poland. Bull. Pol. Ac. Sci. Biol. Sci. 41,
377-392.
Blaszkowski J. 1994.
Arbuscular fungi and mycorrhizae (Glomales) of the Hel Peninsula, Poland.
Mycorrhiza 5, 71-88.
Blaszkowski J., Tadych
M., Madej T. 2002. Arbuscular mycorrhizal fungi (Glomales, Zygomycota) of
the Bledowska Desert, Poland. Acta Soc. Bot. Pol. 71, 71-85.
Blaszkowski J., Tadych
M., Madej T., Adamska I., Iwaniuk A. 2001. Arbuscular mycorrhizal fungi (Glomales,
Zygomycota) of Israeli soils. Mat. II Polsko-Izraelskiej Konf. Nauk. nt. „Gospodarowanie
zasobami wodnymi i nawadnianie roslin uprawnych”. Przeglad naukowy Wydz.
Inz. Ksztalt. Srod. 22, 8-27.
Giovannetti M. 1985.
Seasonal variations of vesicular-arbuscular mycorrhizas and Endogonaceous
spores in a maritime sand dunes. Trans. Br. Mycol. Soc. 84, 679-684.
Hetrick B. A. D., Bloom
J. 1983. Vesicular-arbuscular mycorrhizal fungi associated with native tall
grass prairie and cultivated winter wheat. Can. J. Bot. 61, 2140-2146.
Iwaniuk A., Blaszkowski J. 2004. Arbuscular fungi and mycorrhizae of agricultural soils of the Western Pomerania . Part II. Distribution of arbuscular fungi. Acta Mycol. 39(2), 3-18.
Koske R. E. 1987. Distribution
of VA mycorrhizal fungi along a latitudinal temperature gradient. Mycologia
79, 55-68.
Koske R. E., Walker C.
1986. Species of Scutellospora (Endogonaceae) with smooth-walled
spores from maritime sand dunes: two new species and a redescription of the
spores of Scutellospora pellucida and Scutellospora calospora.
Mycotaxon 27, 219-235.
Miller D. D., Domoto
P. A., Walker C. 1985. Mycorrhizal fungi at eighteen apple rootstock plantings
in the United States. New Phytol. 100, 379-391.
Morton J. M. 1995. Taxonomic
and phylogenetic divergence among five Scutellospora species based
on comparative developmental sequences. Mycologia 87, 127-137.
Morton J. B. 2000. International
Culture Collection of Arbuscular and Vesicular-Arbuscular Mycorrhizal Fungi.
West Virginia University.
Nicolson T. H., Gerdemann
J. W. 1968. Mycorrhizal Endogone species. Mycologia 60, 313-325.
Nicolson T. H., Schenck
N. C. 1979. Endogonaceous mycorrhizal endophytes in Florida. Mycologia 71,
178-198.
Rose S. 1988. Above
and belowground community development in a maritime sand dune ecosystem. Plant
and Soil 109, 215-226.
Tadych M., Blaszkowski
J. 2000. Arbuscular fungi and mycorrhizae (Glomales) of the Slowinski National
Park, Poland. Mycotaxon 74, 463-483.