DEVELOPMENT OF SPORES AND CHARACTERS OF MYCORRHIZAE
OF THE GENUS GLOMUS
In PVLG |
|||
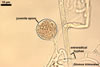
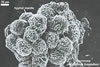
The surface of spores of Glomus spp. may be smooth (in most species) or differently ornamented (e. g., Gl. multiforum Tadych & Blaszk., Gl. pustulatum Koske et al.; Blaszkowski 1994a; Blaszkowski and Tadych 1997; Koske et al. 1986b). Some species produce spores enveloped in a hyphal mantle consisting of interwoven (e. g., Gl. mortonii Bentiv. & Hetrick, Gl. fuegianum (Speg.) Trappe & Gerd.; Bentivenga and Hetrick 1991; Blaszkowski et al. 1998) or dichotomously branched (Gl. corymbiforme Blaszk.; Blaszkowski 1995) hyphae.
In water
|
SEM |
|
Although fungi of the phylum Glomeromycota, except for Geosiphon pyriformis (Kütz.) Wettst. emend. Schüßler, are considered to form obligatorily arbuscular mycorrhizae in mutualistic associations with living plants (Morton and Benny 1990; Schüßler et al. 2001), the formation of spores associated with both mycorrhizal roots and non-root habitats by, e. g., Gl. fuegianum (Blaszkowski et al. 1998; Pegler et al. 1993), Gl. fasciculatum (Thaxt.) Gerd. & Trappe emend. C. Walker & Koske, and Gl. sinuosum (Gerd. & B.K. Bakshi) R.T. Almeida & N.C. Schenck (Gerdemann and Trappe 1974; Koske, pers. comm.) indicate that some species are facultative symbionts (Blaszkowski et al. 1998).
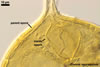
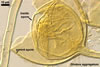
All members of the genus Glomus form spores with only one wall including at least two layers, of which the structural layer consists of many sublayers (laminae). The outer layer or layers adherent to the laminate layer frequently slough(s) with age. In some species, e. g., Gl. claroideum N.C. Schenck & G.S. Sm. and G. etunicatum W.N. Becker & Gerd., the outermost layer stains red in Melzer’s reagent (Stürmer and Morton 1997). A few species, e. g., Gl. claroideum, Gl. lamellosum Dalpé et al. and Gl. gibbosum, produce an inner flexible layer developing from the subtending hypha; this layer frequently is adherent to the inner surface of the laminate layer and, therefore, it usually is difficult to see (Blaszkowski 1997; Blaszkowski et al. 2002; Dalpé et al. 1992; Stürmer and Morton 1997). Of the described species, the inner flexible spore wall layer of only Gl. lamellosum stains in Melzer’s reagent (Blaszkowski et al. 2002); however, this property also occurs in some other undescribed species (Blaszkowski, pers. observ.).
In PVLG |
|||
The wall layers of a subtending hypha are continuous with spore wall layers. During ontogeny, they differentiate at the same time and rate. At the end of spore development, the lumen of the subtending hypha usually becomes closed by either (1) a curved septum continuous with the innermost lamina of the laminate spore wall layer, (2) an invaginated flexible innermost layer, (3) an amorphous plug, (4) thickening subtending hyphal wall, or (5) a combination of the structures mentioned.
In PVLG |
||
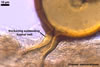
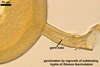
Spores of the genus Glomus germinate by emergence of the germ tube through either the lumen of the subtending hypha (most species) or the spore wall. Germination by both the lumen and spore wall was also observed.
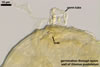
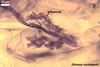
The mycorrhizae of Glomus spp. consist of arbuscules, vesicles (not always formed), and intra- and extraradical hyphae. Arbuscules have cylindrical or slightly flared trunks with branches progressively tapering in width toward tips. Vesicles usually are thin-walled and ellipsoid. Intraradical hyphae usually spread along roots and frequently form Y-shaped branches, H-shaped connections, and coils. Coils mainly occur at entry points.
In roots of Plantago
lanceolata L. |
|||||
REFERENCES
Almeida R. T., Schenck N. C. 1990. A revision of the genus Sclerocystis (Glomaceae, Glomales). Mycologia 82, 703-714.
Bentivenga S. P., Hetrick B. A. D. 1991. Glomus mortonii sp. nov., a previously undescribed species in the Glomaceae isolated from the tallgrass prairie in Kansas. Mycotaxon 42, 9-5.
Blaszkowski J. 1988. Four new species of the Endogonaceae (Zygomycotina) from Poland. Karstenia 27, 37-42.
Blaszkowski J. 1991. Polish Endogonaceae. IX. Glomus aggregatum with spores forming an evanescent outermost wall. Crypt. Bot. 2/3, 130-135.
Blaszkowski J. 1994. Polish Glomales 11. Glomus pustulatum. Mycorrhiza 4, 201-207.
Blaszkowski J. 1995. Glomus corymbiforme, a new species in Glomales from Poland. Mycologia 87, 732-737.
Blaszkowski J. 1997. Glomus gibbosum, a new species from Poland. Mycologia 89, 339-345.
Blaszkowski J., Adamska I., Madej T. 2002. Glomus lamellosum (Glomales, Zygomycota), an arbuscular mycorrhizal fungal species new for Poland and Europe. Mycotaxon 81, 281-292.
Blaszkowski J., Madej T., Tadych M. 1998. Entrophospora baltica sp. nov. and Glomus fuegianum, two species in the Glomales from Poland. Mycotaxon 68, 165-184.
Blaszkowski J., Tadych M. 1997. Glomus multiforum and G. verruculosum, two new species from Poland. Mycologia 89, 804-811.
Blaszkowski J., Tadych M., Madej M. 2000. Glomus minutum, a new species in Glomales (Zygomycetes) from Poland. Mycotaxon 76, 187-195.
Dalpé Y., Koske R. E., Tews L. L. 1992. Glomus lamellosum sp. nov.: a new Glomaceae associated with beach grass. Mycotaxon 43, 289-293.
Declerck S., Cranenbrouck S., Dalpé Y., Grandmougin-Ferjani A., Fontaine J., Sancholle M. 2000. Glomus proliferum sp. nov.: a description based on morphological, biochemical, molecular and monoxenic cultivation data. Mycologia 92, 1178-1187.
Gerdemann J. W., Trappe J. M. 1974. The Endogonaceae in the Pacific Northwest. Myc. Memoir 5, 1-76.
Koske R. E. 1985. Glomus aggregatum emended: A distinct taxon in the Glomus fasciculatum complex. Mycologia 77, 619-630.
Giovannetti M., Avio L., Salutini L. 1991. Morphological, cytochemical, and ontogenetic characteristics of a new species of vesicular-arbuscular mycorrhizal fungus. Canad. J. Bot. 69, 161-167.
Koske R. E. 1985. Glomus aggregatum emended: A distinct taxon in the Glomus fasciculatum complex. Mycologia 77, 619-630.
Koske R. E., Gemma J. N., Olexia P. D. 1986. Glomus microaggregatum, a new species in the Endogonaceae. Mycotaxon 26, 125-132.
Koske R. E., Friese C., Walker C., Dalpé Y. 1986. Glomus pustulatum: A new species in the Endogonaceae. Mycotaxon 26, 143-149.
Morton J. B., Benny G. L. 1990. Revised classification of arbuscular mycorrhizal fungi (Zygomycetes): a new order, Glomales, two new suborders, Glomineae and Gigasporineae, and two new families, Acaulosporaceae and Gigasporaceae, with an emendation of Glomaceae. Mycotaxon 37, 471-491.
Pegler D. N., Spooner B. M., Young T. W. K. 1993. British truffles. A revision of British hypogeous fungi. Royal Botanic Gardens, Kew. 216 pp. + 26 plates.
Schenck N. C., Smith G. S. 1982. Additional new and unreported species of mycorrhizal fungi (Endogonaceae) from Florida. Mycologia 74, 77-92.
Schüßler A., Schwarzott D., Walker C. 2001. A new fungal phylum, the Glomeromycota: phylogeny and evolution. Myc. Res. 105, 1413-1421.
Stürmer S. L., Morton J. B. 1997. Developmental patterns defining morphological characters in spores of four species in Glomus. Mycologia 89, 72-81.